How to calculate ppm of co2 in room
Ĭarbon dioxide was the first gas to be described as a discrete substance. Carbon dioxide also causes ocean acidification because it dissolves in water to form carbonic acid. Since the Industrial Revolution anthropogenic emissions – primarily from use of fossil fuels and deforestation – have rapidly increased its concentration in the atmosphere, leading to global warming. Ĭarbon dioxide is the most significant long-lived greenhouse gas in Earth's atmosphere. It is a feedstock for the synthesis of fuels and chemicals. The frozen solid form of CO 2, known as dry ice, is used as a refrigerant and as an abrasive in dry-ice blasting. It is added to drinking water and carbonated beverages including beer and sparkling wine to add effervescence. It is a versatile industrial material, used, for example, as an inert gas in welding and fire extinguishers, as a pressurizing gas in air guns and oil recovery, as a chemical feedstock and as a supercritical fluid solvent in decaffeination of coffee and supercritical drying. It is an unwanted byproduct in many large scale oxidation processes, for example, in the production of acrylic acid (over 5 million tons/year). It is produced by combustion of wood, peat and other organic materials and fossil fuels such as coal, petroleum and natural gas. Carbon dioxide is produced during the processes of decay of organic materials and the fermentation of sugars in bread, beer and wine making. It is returned to water via the gills of fish and to the air via the lungs of air-breathing land animals, including humans. Since plants require CO 2 for photosynthesis, and humans and animals depend on plants for food, CO 2 is necessary for the survival of life on earth.

In turn, oxygen is consumed and CO 2 is released as waste by all aerobic organisms when they metabolize organic compounds to produce energy by respiration. Plants, algae and cyanobacteria use energy from sunlight to synthesize carbohydrates from carbon dioxide and water in a process called photosynthesis, which produces oxygen as a waste product.
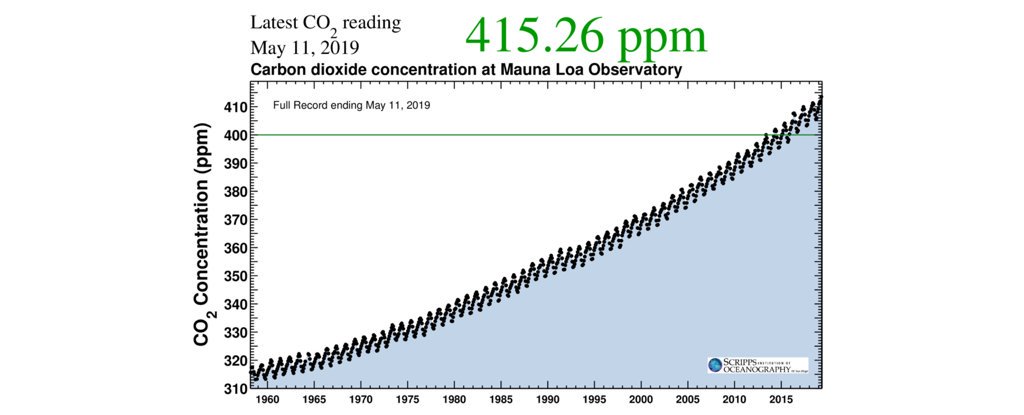
Īs the source of available carbon in the carbon cycle, atmospheric carbon dioxide is the primary carbon source for life on Earth and its concentration in Earth's pre-industrial atmosphere since late in the Precambrian has been regulated by photosynthetic organisms and geological phenomena. However, at normally encountered concentrations it is odorless. Carbon dioxide has a sharp and acidic odor and generates the taste of soda water in the mouth. It is present in deposits of petroleum and natural gas. Because carbon dioxide is soluble in water, it occurs naturally in groundwater, rivers and lakes, ice caps, glaciers and seawater. Natural sources include volcanoes, forest fires, hot springs, geysers, and it is freed from carbonate rocks by dissolution in water and acids. The current concentration is about 0.04% (412 ppm) by volume, having risen from pre-industrial levels of 280 ppm. It occurs naturally in Earth's atmosphere as a trace gas. Carbon dioxide molecules consist of a carbon atom covalently double bonded to two oxygen atoms. 2) is an acidic colorless gas with a density about 53% higher than that of dry air.
